  Additional ResourcesĪs always, if you have any questions, leave a comment below. This gives us C 2H 10N 2.Īnd that’s it! Follow the steps, practice, and you’ll be able to solve these types of problems with no problem. Next, multiply all the subscripts in CH 5N by 2.Divide 62.11 by 31.0571 and this will give you 2. First determine the molar mass of the empirical formula, which is 31.0571 g/mol.If the molar mass of the unknown compound is 62.11 g/mol, what is the molecular formula. Let’s continue with the example we were working through. This will give you the molecular formula. 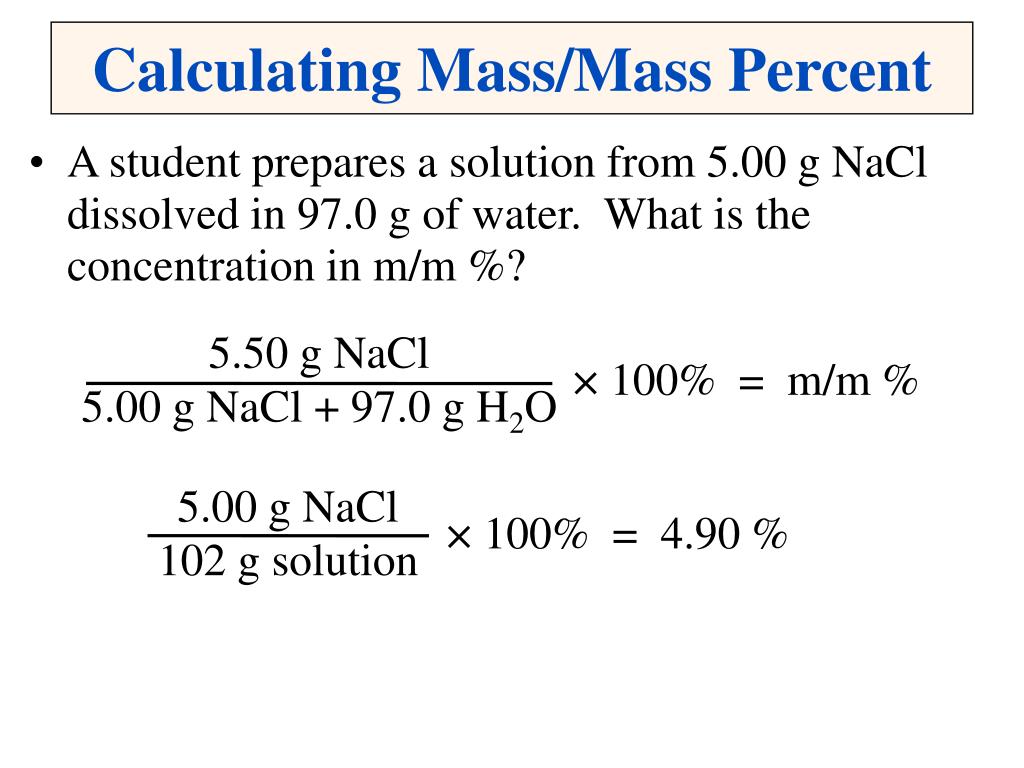
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
